Document Type : Original Research Article
Authors
Department of Chemistry, College of Sciences, University of Baghdad, Baghdad, Iraq
Abstract
Osteoporosis is a long-term degenerative condition that develops when there is an imbalance between bone growth and resorption. Oxidative stress (OS) parameters like total antioxidant status (TAS), total oxidant status (TOS), oxidative stress index (OSI), and malondialdehyde (MDA) are believed to play an important role in this disease. While the two forms of xanthine oxidoreductases enzyme (XDR): xanthine oxidase (XO) and xanthine dehydrogenase (XDH) are parts of the molybdenum iron-sulfur flavin hydroxylase group of enzymes. The purpose of this study is to know the impact of XO, XDH, and OS on this silent disease. Total of (150) samples of postmenopausal women were included in the present study (divided equally to 3 groups as follow: apparently healthy as control, osteopenia, and osteoporosis groups). With the two enzymes, other OS parameters were measured by colorimetric tests. Results showed significantly increase in XO activity in two patients groups compared with control group. In contrast, the XDH activity was significantly decreased. Also, TOS, OSI, and MDA were significantly increased while TAS was significantly decreased between the groups of patients compared with the control group. Based on the observations, it can be concluded that such an increase XO activity is due to the increase conversion of the other form (XDH) to XO led to the production of reactive oxygen species and so, OS increase. The present data provide further evidence of the relation of such disease with OS and the role of XOR generation of oxidant molecules and inflammatory reaction in this common disease.
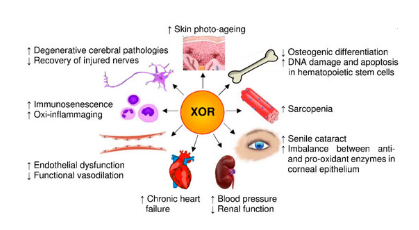
Graphical Abstract
Keywords
Main Subjects
Introduction
Osteoporosis (OP) is a chronic skeletal condition characterized by a loss of bone mass and bone mineral density (BMD). As a result, the quality or structure of bone deteriorates, weakening the bone structure and perhaps causing fractures. Due to the absence of noticeable defects, patients frequently remain oblivious that their condition is progressing until a fall or other impact with sufficient force results in a bone fracture [1]. The OP disease that shows a reduction in bone mass, micro architectural disruption, and enhanced skeletal fragility, with subsequent low bone strength and high rates of fracture [2]. The disease causes bones to deteriorate in density, become brittle, and cracks more easily [3]. Measurements of bone turnover markers (BTMs) and BMD can be used to forecast it since an increase in BTMs is related to a decrease in BMD. It happens when bone resorption surpasses bone production [4]. As a result, identifying people who are at a higher risk of fracture is critical for the early intervention and lowering fracture risk due to the rapid expansion of the older population, OP has now become an epidemic in several countries [5]; demographic transition in developing countries i.e. prolonged life expectancy might increase the prevalence of OP [6]. More than 9 million fractures occur each year due to OP worldwide, meaning that a fragility fracture occurs every three seconds [7]. OP affects about one in every four women, its prevalence increases with age from 2% at 50 to >25% at 80 years old, which reflects the significant increase in bone loss rate in postmenopausal women after losing the protective effect of estrogen. Women from Africa and the Caribbean tend to be less vulnerable than white or Asian women [8]. Disease acuteness and bone mass loss are linked to increase inflammation-stimulating cytokines systemic inflammatory disorders [9]. Meanwhile, OP is related with oxidative stress (OS) which produced when there is an imbalance between the formation of reactive oxygen species (ROS) as oxidants and the capacity of antioxidants to scavenge them [10,11]. The produced ROS caused osteoblasts to apoptosis and reduce their activity, which was also detrimental to osteogenesis. Furthermore, the high levels of ROS block and reduce osteoblast differentiation, resulting in decreased mineralization and osteogenesis [12]. Likewise, chronic inflammation, aging, and OP can all be caused by an excess of free radical damage to biomolecules [13].
Xanthine oxidoreductase (XOR) is a housekeeping enzyme that is a member of the molybdenum iron-sulfur flavin hydroxylases. It plays a major role in the catabolism of purines by catalyzing the two final stages that result in the formation of uric acid from hypoxanthine and xanthine [14]. It is widely distributed in the heart, liver, kidney, and vascular endothelium [15]. The enzyme only has dehydrogenase activity in organisms, but it has two interchangeable activities in mammals: xanthine dehydrogenase (XDH, EC 1.17.1.4) and xanthine oxidase (XO, EC 1.17.3.2) [16] in which XDH reduces NAD+ to NADH, while oxygen is used by XO to generate hydrogen peroxide and superoxide [17-19]. According to the points mentioned above, the present study aimed to evaluate the XOR activities and OS status in serum samples of postmenopausal Iraqi women.
Materials and methods
This study has been performed in the period from October to December 2021 including 150 postmenopausal Iraqi women who were attending Baghdad Teaching Hospital, in the Medical City Complex, Iraq. The women were divided into three groups according to the T-score values for BMD obtained from the dual-energy X-ray absorption spectroscopy DEXA scan. The first group included 50 apparently healthy individuals (age 59 ± 7.05). The second group included 50 patients with osteopenia (age 59.02 ± 7.41), while the third group consisted of 50 patients with osteoporosis (age 59.1 ± 7.65). The body mass index (BMI; Kg/m2) was measured by weight in (Kg) / length in (m2). All participants were subjected to a personal interview by using especially designed questionnaire format full history with detailed information after getting their approval. The exclusion criteria of samples included diabetes mellitus, rheumatoid arthritis, hypothyroidism, and hyperthyroidism-patients that were treated with steroid drugs. Protocol was approved by the Ethics Committee of the College of Science/ University of Baghdad. Samples collection included 10 mL of venous blood that were placed in the tube at room temperature and centrifuged for 5 minutes at 3,000 cycles/min. The serum was put into an eppendorff tube and stored in -20℃ freezer until it was analyzed. The XO activity was determined by the method of Ackermann and Brill, while the XDH activity was determined by Fried et al. method. The MDA measurement was done by using Satoh modified method. Both TOS and TAS levels in serum were determine by using Erel’s methods. The findings were presented as (micromolar hydrogen peroxide equivalent per liter; μmol H2O2 Eq./L) values of TOS and (millimolar uric acid per liter; mmol uric acid Eq./L) values of TAS. The following equation [14] was used to calculate the OSI value: OSI = TOS (µmol H2O2 Eq./L) / TAS (μmol glutathione Eq./L).
The data were fully characterized using SPSS by licensed materials version 26 computer software. The data in this study were presented as a Mean± Standard deviation (Mean± SD) by using One-way ANOVA. ROC is a statistical analysis that uses a plot of the association between sensitivity and 1-specificity to determine a diagnostic test's optimal specificity and sensitivity [20].
Results
The study included one hundred and fifty Iraqi women, the mean ± SD for their age, BMI, BMD, and T-score were represented in (Table 1). Depending on this information, the three groups were classified, and the results indicated significant variations in BMD and T-score between them.
A person's BMI is an anthropometric measurement based on their weight and height that aids in assessing their obesity status, as reported in Table 1. There was a non-significant difference in age and BMI (p =0.977) between the studied groups that were obese people. There was a non-significant difference in (Mean ± S.D) age between the studied groups.
The activities of both XO and XDH were summarized in Table 2. The results appeared to show significant increases between XO activities of Osteopenia and OP when compared with control. While the XDH activities of both patients groups were decreased comparing with control group, and the OP decreased significantly. It is clear that OP patients revealed the higher mean value of XO activity than osteopenia patients on the other hand, the XDH activity was lower, but the results were non-significant.
Coloremetric methods were used to evaluate the OS status in the sera of the three studied groups, as illustrated in Table 3.
Indicators of OS (TOS and OSI) were in a highly significant increase (p<0.001) and MDA (p˂0.01) in were significantly increased in patients group compared with the control groups. The result indicates a high significant change in patients group compared with the control groups in the TAS (P< 0.001), as listed in Table 3. The OSI value increased significantly between the two patients groups, while MDA was not significantly higher in OP group.
The correlation coefficient between XO and XDH with individual parameters in control group is shown in Table 4.
The correlation coefficient between XO and XDH with patient's parameters in osteopenia group is demonstrated in Table 5.
The correlation coefficient between XO and XDH with patient's parameters in Osteoporosis group is shown in Table 6.
ROC curves for XO, XDH, and OSI to control and osteopenia, control and osteoporosis, respectively, are depicted in Chart 1.
Discussion
The silent disease (OP) is characterized by low bone mass as a result of bone loss and insufficient bone production. This causes bones to become more brittle, increasing the likelihood of fractures, particularly in the hip, spine, wrist, and shoulder regions [3]. Age is a risk factor. In several diseases especially in OP, the decline in the bone marrow and osteoblast is linked to age-related alterations in creating and distinguishing [21]. One in every three women over the age of 50 and one in every two women over the age of 60 will experience an OP-related fracture. The results of (Table 1) show no significant change between three groups. Hammed EO, et al.[22] and Zavar-Reza J. [23] results appeared that no difference in the age of patients compared with control, confirming our findings. The reason for this is that the study was conducted on a specific age group and fixed for all groups. The age (mean ± S.D) of OP, Osteopenia and control groups were (59.1 ± 7.654), (59.02 ± 7.413), and (59 ± 7.057) years, respectively. While, the participant women in this work were aging and postmenopausal. The current study showed that there was no significant difference in BMI between patients and controls, as indicated in Table 1. However, all participants in this wok were obese. Obesity is a worldwide health problem. When the BMI exceeds 30 kg/m2, the word "obesity" is used, it affected 600 million people and 42 million children under five in 2014. According to several studies, this is the most severe threat to human life, and it is more prevalent in women, due to the common progenitor stem cell's abnormal commitment to adipocytes rather than osteoblasts observed in obesity, increased bone marrow lipid metabolism may decrease bone mass in obese patients [24]. According to recent studies, an increase in BMI is harmful to BMD and bone formation [25]. They only discovered a delay in bone loss in obese women to a delayed x-ray response by fat [26]. The XOR a key enzyme in the breakdown of purines, catalyzing the conversion of xanthine, and hypoxanthine into the final metabolite, urate.The current study found significant increases in the serum XO activities in Osteopenia and OP compared with the control group, with a significant decrease in serum XDH activities in patients with OP compared with control group (Table 2), the results were consistent with other studies on another inflammation diseases which indicate that XO levels are increased in several pathological states like inflammation [27]. The fluctuation in the results of osteopenia between the control group and the OP group may be due to a T- score values that may be near that of control, while its value for OP was about two fold that of osteopenia. The XOR can promote superoxide formation in low pH and hypoxia. Although different enzymes are involved in generating these species. The XOR activities may produce a variety of ROS and RNS [28]. XOR and its products can induce inflammation and OS [16]. Superoxide produced by xanthine and XO systems promotes new osteoclast production and bone resorption, suggesting that free oxygen radicals produced in the bone can promote osteoclast formation. Furthermore, hydrogen peroxide induces the production of osteoclasts in a concentration-dependent way, increasing bone resorption [29]. An imbalance between pro-oxidants and antioxidants, which is caused by higher levels of (RNS) and/or (ROS), is what the OS concept refers to as a decrease in antioxidant defense systems[30]. Systems [30]. Several studies have demonstrated that OS decreases bone formation by reducing the differentiation and survival of osteoblasts. However, while ROS can activate osteoclasts, they can also boost bone resorption [31]. This intracellular OS promotes osteoclast bone resorption associated with inflammatory cytokines while inhibiting osteoblast bone growth [29]. XOR may play a role in the OS pathophysiology and the inflammatory response produced by uric acid and XOR-derived reactive oxygen radicals[28]. Radicals [28]. OS is produced when there is an imbalance between the ROS formation and the capacity of antioxidants to scavenge them [10]. By increasing the inflammatory response and modifying the survival and differentiation of osteoblasts, OS accelerates bone remodelling turnover and bone mass loss. In contrast, antioxidants reduce OS and bone mass loss [32,33]. The results of (Table 3) revealed that the oxidants (or their markers MDA) increased gradually from control group through osteopenia to OP and vice versa for antioxidants in spite of that some results were not significant. ROS has been suggested to play a major role in OP including bone resorption, with osteoclast-generated superoxide contributing directly to bone degeneration. As a result, the clinical evaluation of TAS and OSI was explored [10]. A study in China found that OP was correlated with a significant decrease in TAS and an increase in OSI, suggesting that the formation of OP is associated with an imbalance of ROS and antioxidant systems. The results further verified the significance of OS and ROS in the development and progression of osteoporosis [10].Also, a study in Turkey by Altindag O. et al. found lower TAS, higher TOS, and a higher OSI in OP patients than healthy controls [34].This is consistent with our study. MDA is one of the terminal products of intracellular polyunsaturated fatty acid peroxidation. An increase in unfastened radicals causes overproduction of MDA [35]. MDA levels in OP women were significantly higher than in healthy individuals [35, 36]. The current results approved these studies.
The correlation results in Table 4 indicate a negative correlation between XDH and TOS, but a positive correlation between XDH and TAS in osteopenia patients. In OP patients, XO has a negative correlation with XDH, as illustrated in Table 5. The XOR serum levels are usually very low in humans. However, XOR levels may become more elevated in some pathological conditions, which cause XOR release from damaged cells into the circulation, where it is converted into the oxidase form [37] .XO is produced when the native form of XDH is altered either by sulphydryl oxidation or by limited proteolysis [38]. XDH antioxidant enzymes is responsible for maintaining the balance between the functions of free radical formation and eliminating their excessive amounts[39]. The storage of organ antioxidants, primarily from the liver and adipose tissues, as well as the stimulation, or activation of antioxidant enzymes prevent TAC deficiency caused by oxidative stress. The levels of these antioxidants are suitable not only as a protection against oxidation, but also reflect their consumption during acute oxidative stress state [40]. The results of the Roc curve showed the diagnostic susceptibility of XO and OS to osteopenia and OP.
Conclusion
Based on the obtained findings, due to significant differences between the study groups, the XOR enzymes play an important role in the OP development. Increased XO activity is due to the increase conversion of the other form (XDH) into XO causing production of reactive oxygen species, and so increasing OS. In addition, we can conclude that the OS status in OP Iraqi woman plays a key role in the development of osteopenia, osteoporosis, and its complications. Moreover, the OS parameters such as the TAS decrease and increase of TOS, OSI, and MDA appear to be among those that predict long-term complications. According to the ROC curve results, XO and OSI may help diagnose postmenopausal women with Osteopenia and OP. These data may support future strategies for the OP prevention where the control of such markers may have therapeutic value.
Acknowledgments
The authors would like to express their deep thanks to all the staff in the Osteoporosis Department and all the workers in the Emergency laboratory at Baghdad Teaching Hospital in the Medical City Complex, Iraq. The authors would also like to thank all of the participants who provided information and blood samples for this study.
Orcid:
Noor Abdulhussain Naama: https://orcid.org/0000-0002-4707-7289
-----------------------------------------------------------------------------
How to cite this article: Noor Abdulhussain Naama, Jwan Abdulmohsin Zainulabdeen. Evaluation of oxidative status with xanthine oxidoreductase activities in postmenopausal Iraqi osteoporosis women. Eurasian Chemical Communications, 2022, 4(12), 1295-1305. Link: http://www.echemcom.com/article_154190.html
-----------------------------------------------------------------------------
Copyright © 2022 by SPC (Sami Publishing Company) + is an open access article distributed under the Creative Commons Attribution License(CC BY) license (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
