Document Type : Original Research Article
Author
Baquba Technical Institute, Middle Technical University, Baghdad, Iraq
Abstract
In the present study, nanoparticles of copper oxide were formed using a green approach. The Biosynthesis of CuO NPs was fabrication with the help of fungi which isolated from rot vegetables. The extract of Aspergillus niger fungi and Copper(II) chloride were used to synthesize bio-CuO NPs. Different techniques such as XRD (X-ray diffraction), FE-SEM (Field emission scanning electron microscopy), FTIR (Fourier transform infrared spectroscopy), and UV-Vis (UV–visible spectroscopy) were utilized to characterize and identify the biosynthesis of CuO NPs. The bioactivity of bio-CuO was screened against some types of pathogenic bacteria including Staphylococcus aureus, Bacillus subtilis, Escherichia coli, and Salmonella typhimurium via MHA (Muller Hinton agar) media. The results show that bio-CuO NPs have a potential effect against bacteria used in the current study and are more active toward Escherichia coli and Salmonella Typhimurium compared to other types. The antioxidant activity of bio-CuO was further studied in a dose-dependent manner. The obtained results show a positive effect of bio-CuO NPs and the activity increase when concentration is increased. Therefore based on the obtained results, the bio-CuO NPs have promising applications in the field of biomedical, industrial, and biological applications.
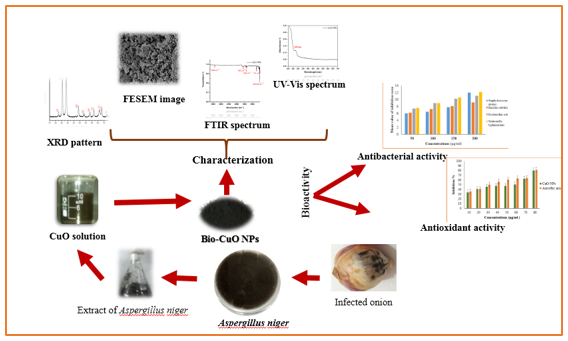
Graphical Abstract
Keywords
Main Subjects
Introduction
Recently, nanotechnology has been considered as one of the most widely used technologies in most fields and applications. Where they can be used in the field of industry, energy, medicine, agriculture, cosmetics, and many other applications, this technology gained its importance and advantages due to the large surface area to volume ratio, which gave it many benefits that help it to use in various fields. With the progress of science and research, the progress and development of nanotechnology and its uses increased which often had a positive impact on the comfort and well-being of humanity [1]. Nanoparticles, especially metal oxide nanoparticles, are of great and wonderful importance that contributes to many applications because of their physical, chemical, magnetic, and other characteristics that help and contribute to their use for the benefit of humanity, the environment, food, health, and others [2]. Among metal oxide nanoparticles, are copper oxide nanoparticles, where copper is considered one of the promising elements characterized by good qualities that make it possible to use many applications. In addition, when copper is linked to oxygen, is copper oxide formed (p-type semiconductors) and possessed an energy band gap (≈1.7) eV [3,4], which increases its use in several fields. When it is converted into nanoparticles, the nanoparticles of copper oxide become characterized by unique features that make them subjected to attention to be used and benefit from its advantages. Nano copper oxide has physical, chemical, electrical, optical, thermal, and many other properties that make it different from bulk material [5, 6]. It is a possession of these distinctive features facilitated its use in a wide range of applications, including catalysts [7], gas sensors [6], water pollutant removal [8], solar cells [9], and antimicrobials, anticancer [10,11] as well as many other important and useful applications summarized in reviews [12,13].
Copper oxide nanoparticles are formed by several physical or chemical methods including microwave radiation, sol-gel, thermal decomposition, solid-state reactions, precipitation, co-precipitation, and many other methods [14-17]. Each of these methods has some advantages and disadvantages, but the disadvantages and drawbacks of using these methods are more than the advantages. This is due to its reliance on materials that may be harmful to users in addition to its harm to the environment, dependence sometimes on high pressure and temperature, energy consumption is sometimes large, and other disadvantages or drawbacks [18,19]. Therefore, researchers tend to prepare nanoparticles in a way that is environmentally friendly, harmless, and simple approach. The extracted plants, fungi, algae, or other green methods that rely on in the preparation of these nanoparticles and used as a reducing agent of ions metal. There are many researchers that focused on the use of plant extracts in preparation, and they elaborated extensively in this direction, Akintelu et al. explained in detail in their review [18]. Using fungi in the preparation of nanoparticles does not receive much interest and research. Therefore, we focused our current research on the formation of copper oxide using fungi. Due to fungi having the capacity to prepare a variety of NP (nanoparticles) and MONPs (metal oxide nanoparticles) types through intracellular and extracellular pathways as well as quick mycelia growth, which provides a larger surface area and easy handling of biomass. Likewise, their capacity to secrete large amounts of proteins help increase the productivity of NPs and MONPs creation. Fungi have been found to be great candidates for the synthesis of NPs and MONPs in nanotechnology with a variety of potential uses in biomedicine and industry [20-22]. In the current work, Aspergillus niger is used to synthesize CuO NPs that are isolated from root vegetables. The CuO biosynthesis is subjected to an antibacterial test and antioxidant screening.
Materials and methods
Materials
Copper(II) chloride (CuCl2,99%), Potato Dextrose Agar (PDA), Potato Dextrose Broth (PDB), and Whatman filter paper all pushed from Sigma Aldrich-Germany.
Isolation fungi and extraction
An infected onion sample was collected and inoculated in Petri plates that contain PDA (potato dextrose agar) at the temperature of 28±2 °C and the appropriate conditions to grow for 7 days. Aspergillus niger was grown and the fungi colony was identified using optical microscopy and macroscopic features such as diameter, conidial color, colony reverse, smoothness, and monitoring growth rate. The obtained results were compared to Raper et al. [23]. The obtained Aspergillus niger was again cultured in PDB (Potato Dextrose broth) that contain Glucose, 3 gm, Ampiclox antibiotic, 3 mL, and 300 mL of PDB was kept in suitable condition in the dark room at 28 ± 2 °C, with continued shaking (150 rpm) for one week under observation. After the incubation time, the biomass of fungi was harvested via filtration by using filter paper (Whatman No.1). After that, the filtrate was washed three to four times using sterile distilled water. The filtered mycelia biomass was added to sterile flask contain 100 ml of sterile distilled water and kept under shaking at 150 rpm for 3 hrs at room temperature. The biomass of fungi was harvested via filtration by using filter paper (Whatman No.1). The final solution of A. niger extract was used for the biosynthesis of CuO NPs [20,24,25].
Biosynthesis of copper oxide nanoparticles
To prepare biosynthesis of CuO, the solution CuCl2 (3 mM/50 mL) was added to A. niger extract filtrate solution (50 mL) and subjected to continuous stirring at 50 C for 4 hrs with monitoring pH (10-11). The color of the solution changed to brown which indicated the formation of CuO NPs. The final solution was centrifuged at 15,000 rpm for 20 min, and after that washed using ethanol with sterile distilled water, and then dried, crushed, and annealed at 400 C for 2 hrs. [26], and kept it for further characterization
Characterization of biosynthesis copper oxide nanoparticles
X-ray diffractometer (BRUKER D2) with (λ=0.15405 nm) and Cu-K radiation was used as a source. Morphology of bio-CuO NPs was carried out using a Field Emission Scanning Electron Microscope (FESEM-FEI Nova Nano SEM 450). Fourier Transform Infrared FTIR spectroscopy (Shimadzu) was used to find out the functional groups in the range between 400 and 4000 cm-1 at room temperature. UV-Visible spectrometer (Jasco UV, V-750) was used to find out the optical properties of bio-synthesized CuO.
Bioactivity of bio-CuO NPs
Antibacterial activity assay
Firstly, we prepared a stock solution of bio-CuO NP in dimethyl sulfoxide (DMSO) media, then we diluted into 4 concentrations, 50 μg/ml, 100 μg/mL, 150 μg/mL, and 200 μg/ml, respectively. Four types of bacteria such as Staphylococcus aureus, Bacillus subtilis, Escherichia coli, and Salmonella typhimurium were used to assay the bioactivity of bio-CuO NPs via the disc diffusion method [27]. The bacteria were swabbed on the Petri plates (90 mm) containing Muller Hinton agar (MHA) media with help of cotton swabs and the sterile filter discs (6 mm) containing different concentrations of bio-CuO were put carefully on the surface of the bacteria and incubation for 24 hrs at 37 °C. After incubation time, the activity of nanoparticles was evaluated by measuring the zone of inhibition (mm) and repeated three times. The mean value was taken and (DMSO) was used as a negative control, whereas Chloramphenicol used as a positive control.
Antioxidant activity assay
2,2-Diphenyl1-picrylhydrazyl (DPPH) radical scavenging was used to assay the antioxidant of biosynthesis CuO NPs as explained in [28,29] with some modification. The different concentrations (10, 20, 30, 40, 50, 60, 70, and 80) μg/ml of bio-CuO NPs were used. The prepared solution was kept in a dark room and shaken for 30 min. The absorption peak of samples was measured using UV-Vis spectrophotometer at λ = 517 nm. At the same time, the positive control (ascorbic acid (AC)) was used. The experiment was repeated three times and the average absorption value was taken. The percentage of antioxidant activity was calculated using Equation (1):
Results and discussion
XRD study
The bio-synthesized CuO NPs using Aspergillus niger were characterized via X-ray diffraction, as depicted in Figure 2. The diffraction peaks are located at 32.6º, 35.5º, 38.6º, 48.8º, 53.4º, 58.5º, 61.6º, 66.3º, 68.1º, and 72.7º corresponded to (110), (-111), (111), (-202), (020), (202), (-113), (311), (220), and (311) planes, respectively, which are consistent with JCPDS file No.05-0661 which confirmed the monoclinic structure bio-CuO NPs. The diffraction pattern shows no additional peaks were observed that indicated the success and the crystalline nature of biosynthesis CuO NPs [30]. Crystallite size of Bio-CuO NPs is calculated with the help of Debye-Scherrer Equation (2).
Where, λ= 1.5418 Å, β indicates to FWHM (full width at half width maximum), θ indicates to the angle of Bragg diffraction, and D is the crystal size of NPs. The average crystal size of bio-CuO NPs was calculated and is found 15 nm.
FESEM study
Figure 3 indicates the morphology of biosynthesis of CuO NPs was analyzed using a FESEM image. It is found from Figure 3 that bio-CuO NPs are arranged and have a spherical-shape with uniform distribution.
FTIR study
The functional groups of biosynthesis CuO NPs were studied with a help of the FTIR technique. Figure 4 depicts the spectrum of bio-CuO NPs. The absorbed peak at 3395 cm-1 is attributed to N–H stretching vibration [11,27], while a peak at 1662 cm-1 is attributed to the C=O bond of an amide group [27]. The absorption peak appears at 1408 cm−1 assigned to C–O stretching, while the absorption peak at 741 cm-1 is assigned to the C-H bond. The peaks in the range 584-500 cm-1 corresponding to metal-oxygen bond (Cu-O) [28] that confirm the success of biosynthesized nanoparticles of CuO.
UV-Vis study
In this investigation, the CuO biosynthesis was confirmed using UV-Vis spectrometer. The high-intensity peak that was absorbed in 278 nm was related to the formation of nanoparticles CuO and it is in agreement with the literature of CuO NPs [31-33]. Figure 5 illustrates the absorption peaks of bio-CuO NPs.
Bioactivity analysis of bio-CuO NPs
Antibacterial activity analysis
The activity of bio-CuO NPs was examined against some types of bacteria. The zone of inhibition was measured after 24 hours in a dose-dependent manner. The results are presented in Table 1 and shown in Figure 6. Bio-CuO possesses excellent activity against different types of bacteria, as explained in different reports [5,34,35,36]. The increased concentrations of samples also lead to an increase in the inhibitory.
Nanoparticles prepared in greenways play a major role in their use as antibacterial, due to the ability of these nanoparticles to penetrate the cell wall and inhibit their work. The nanoparticles mechanism especially copper oxide nanoparticles which used in this study lies in their ability to release some positively charged ions (Cu+2) that accelerate to adhere to the negatively charged bacterial cell wall. This is might due to the electrostatic phenomenon, an attractive force occurs between the released ions and cell wall, which leads to tearing this wall and inhibiting the activity of bacteria and sometimes to their death [34,35]. In addition, the nano-size of nanoparticles which have the ability to penetrate the membrane cell of bacteria (micrometers).
Antioxidant activity analysis
One of the most significant fundamental investigations of nanomaterials is anti-oxidative activity. All life systems depend heavily on antioxidants and free radicals are created in biological systems when biomolecules interact with molecule oxygen [38].
The antioxidant activity (inhibition %) of bio-CuO NPs using DPPH radical is demonstrated in Figure 7. The activity of nanoparticles was studied in a dosing method by increasing the nanoparticles concentration of copper oxide. The % of inhibition shows an increase in the inhibitory as (33.80%, 40.63%, 45.18%, 47.27%, 47.35%, 50.51%, 62.66%, and 80.04%) when the concentration of bio-CuO increment as (10, 20, 30, 40, 50, 60, 70, and 80) μg/ml. From the Figure (7), the ability of bio-CuO to scavenge free radicals can be observed and it is very near to the stander AC [22,37,39]. At the high concentration, the CuO exhibited high activity to scavenge free radicals reach to 81%.
Conclusion
The green methods are simple, low-cost, and eco-friendly methods. Bio-CuO nanoparticles in 15 nm in crystal size with a spherical shape were synthesized using an extract of Aspergillus niger fungi. The formation of bio-CuO NPs was identified via different techniques. Bio-CuO confirmed their activities when examined against some kinds of bacteria and antioxidant tests. Antibacterial property revealed good activity toward bacterial under-investigated. The analysis of antioxidants shows the ability of bio-CuO to free radical scavenging. These bioactivities of bio-CuO NPs make them applicable to use various bio-applications and packaging of food.
Acknowledgements
I would like to give my great thanks to all laboratory technicians who helping me to complete the practical part of my work.
Conflict of Interest
The author declared there is no conflict of interest.
Orcid:
Abdulwadood Ibrahim Arif: https://orcid.org/0000-0002-4299-0722
----------------------------------------------------------------------------
How to cite this article: Abdulwadood Ibrahim Arif*. Biosynthesis of copper oxide nanoparticles using Aspergillus niger extract and their antibacterial and antioxidant activities. Eurasian Chemical Communications, 2023, 5(7), 598-608. Link: http://www.echemcom.com/article_168762.html
----------------------------------------------------------------------------
Copyright © 2023 by SPC (Sami Publishing Company) + is an open access article distributed under the Creative Commons Attribution License(CC BY) license (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
