Document Type : Original Research Article
Authors
Department of Chemistry, College of Science, University of Baghdad, Baghdad, Iraq
Abstract
Polymerization of precipitation was used to make liquid electrodes with metformin (MET) imprinted polymers. lev was used as a template to make molecular (MIP) and non-imprinted (NIP) polymers. In the polymerization process, methyl methacrylate (MMA), acrylamide (AM), ethylene glycol dimethacrylate (EGDMA), and benzoyl peroxide (BPO) were employed as monomers, cross-linkers, and initiators. Using Di B Sabacat (DBS) and Di methyl adipate (DMA) as plasticizers in PVC matrix, the molecularly imprinted membranes and the non- membranes were prepared. The liquid electrodes' slopes and detection limits were -28.03 – -19.45 mV/decade and 3×10-5 M–7×10-5 M, respectively, and their reaction time was about 1 minute. Liquid electrodes were filled with 0.1 M standard drug solution, and their response was consistent throughout a pH range of 1.5 to 10.0, with high selectivity for several types. The electrodes developed have been effectively used in the production of pharmaceutical samples for analyte analysis without the need for time-consuming pretreatment.
Graphical Abstract
Keywords
- Liquid electrodes
- metformine (MET)
- (MIP)
- acrylamide (AM)
- ethylene glycol dimethacrylate (EGDMA)
- benzoyl peroxide (BPO)
Main Subjects
Introduction
Metformin, marketed under the trade names Glucophage and others, is the first-line therapy for type 2 diabetes, particularly in overweight people [1,2,3,4]. It is also used to treat polycystic ovarian syndrome (PCOS) [5]. It is taken by mouth and is not linked to weight gain [6]. It is occasionally used as an off-label supplement to help patients who take antipsychotics and phenelzine to avoid gaining weight [7]. Metformin (Figure 1) is a medication that is typically well tolerated [8]. Diarrhea, nausea, and stomach discomfort are all common side effects. It has a minimal chance of inducing hypoglycemia. If the drug is given in excessively large dosages to people who have significant renal issues, a high blood lactic acid level can occur [9]. It is not advised for people who have severe liver disease. Metformin is a kind of antihyperglycemic drug known as a biguanide. Molecular imprinting is a new approach for creating polymers with distinct molecular characteristics for a specific medication, its analogs, or an enantiomer [10]. Molecularly imprinted polymers (MIPs) are made. In a suitable solvent, a template molecule is combined with functional monomers, a cross-linker, and an initiator [11]. Aprotic and non-polar solvents are used most of the time. The extraction of the template molecule after polymerization exposes recognition holes that complement the template molecule's shape, size, and chemical activity, allowing the resultant polymers to preferentially rebind the template molecule from a mixture of closely similar molecules [12].
For metformin determination, several analytical techniques were employed, including chromatography [10], high-liquid chromatography (HPLC) and molecular absorption, flow injection [11,12], and potentiometric method [13,14]. On the other hand, such procedures are differentiated by the time and effort required to prepare the sample for analysis, as well as the high cost of instrumentation. A novel metformin-selective membrane electrode (MET-MIP+DBS or DMA) for the PVC membrane process was developed in this work.
Experimental
Chemicals and materials
Company of Drug Manufacturing and Medical Supplies provided metformin (IRAQ- SID- Samara). Metformin 850 mg from (Berlin-Chemie AG Berlin-allemagne) and METFORMIN 500 Merck Serono KGaA, Darmstadt from (Germany) are two generic metformin tablets available in local pharmacies. Sigma-Aldrich provided them, and they were utilized as directed. From Sigma-Aldrich, methyl methacrylate (MMA), acrylamide (AM) (99%), monomer ethylene glycol dimethacrylate (EGDMA) (99%), and benzoyl peroxide (BPO) (78%) were used. The chemical used was the largest purity reagent and was used when obtained without further purification.
Apparatus
A digital voltmeter (HANA pH211instrument Microprocessor pH meter) was used to perform potentiometric measurements. Digital pH meters (wissenschaftlich-TechnischeWerkstätten GmbH WTW pH meter in lab pH720-Germany) were used for pH measurements. Electrode efficiency was investigated by calculating the potential of levofloxacin solutions at ambient temperature with a variety of concentrations from 5×10-4 M to 10-1 M. Every solution was stirred, and the reading potential was recorded at equilibrium. The calibration curves were obtained by plotting the response against the levofloxacin concentration logarithmic functions.
Preparing of standard solutions for ISEs studies
1- A standard solution of 0.1 M metformin was prepared by dissolving 1.290 gm standard metformin in methanol and in the volumetric flask completed to 100mL. Other solutions (5×10-5 – 10-1) M were prepared in 100 mL with the same procedure.
2- Methyl prarben (1×10-1 M) solution was prepared by dissolving (1.521 g) of Methyl prarben in 100 mL of deionized distilled water in volumetric flask of 100 mL. A series of solutions has been prepared from (1×10-1 M) of Methyl prarben.
3- Propyl prarben (1×10-1 M) solution was prepared by dissolving (1.802 g) of Propyl prarben in 100 mL of deionized distilled water in volumetric flask of 100 mL. A series of solutions have been prepared from (1×10-1 M) of Propyl prarben.
4- Tri sodium citrate (1×10-1 M) solution was prepared by dissolving (2.5807 g) of Tri sodium citrate in 100 mL of deionized distilled water in volumetric flask of 100mL. A series of solutions has been prepared from (1×10-1 M) of Tri sodium citrate.
5- The stock standard solution of 1×10-3 M, 1×10-4 M, phospho molybdic acid was prepared by dissolving 1.1288 g, 0.11288 g, respectively, in distilled water and completed to 100 mL.
6- Hydrochloric acid (1N and 0.1N) was prepared from (8.36 mL and 0.836 mL) of concentrated HCl, respectively, and was diluted by deionized water to 100 mL.
7- Sodium hydroxide (1N and 0.1N) was prepared from (4 gm and 0.4 gm) of
NaOH respectively and diluted by deionized water to 100 mL.
Synthesis of the imprinted polymer (MIP)
The first molecularly imprinted polymer (MET-MIP1) was formulated using a bulk polymerization technique in a 50mL screw cap glass test tube (50 mL); MIPs for The template (MET) 0.127 mmol (0.016 g) was dissolved in a thick walled glass tube in 2 mL of methanol (CH3OH), 2.5 mmol (0.25) g methyl methacrylate A functional monomer, cross-linker N,N methyleneediacrylamide (N, N-MDAM) 14 mmol (2.15) g and 0.08 mmol (0.01g) benzoyl peroxide as initiator (BPO). But while the second molecularly imprinted polymer metformin (MET-MIP2) was obtained by combining 0.42 mmol (0.05 g) of the template (MET), it was dissolved in a thick-walled glass container in 2mL methanol. 2.7 mmol (0.19) gm acrylamide as a functional monomer, Ethylene glycol dimethacrylate (EGDMA) 12.7 mmol (2.5 g) as across-linker and initiator benzoyl peroxide (BPO) 0.15 mmol (0.036 g). The mixture was degassed in an ultrasonic water bath, purging nitrogen for 40 minutes to remove oxygen from the solution. The glass tube was withdrawn from the ultrasonic water bath while preserving the flow of nitrogen, sealed and put within a water bath at 60 °C to start the reaction. The above solution was later added. The mixture was degassed in an ultrasonic water bath, purging nitrogen for 30 minutes. The glass tube was placed in a water bath at 60 °C while maintaining the continuous flow of nitrogen inside the glass tube throughout the reaction time, when the reaction ended, the molecular imprinted polymer was hardened, the polymer was dried and crashed to acquire a polymer particle after the polymerization process. The template was successively eliminated using Soxhlet removal by continuous washing with the MIPs with 40 percent (v /v) portions of 100 mL methanol /acetic acid solution. The polymer was dried for (24-48) hours at (35-45) °C. The polymers were then crushed with mortar and pestle and measured at a particle size of 125 μm (using 100 mesh sieves); they were used in the selective sensor membrane as an active substrate. The unprinted polymer (NIP) was similarly developed but without the drug template.
FTIR of molecularly imprinted polymers for (MET)
The metformin FTIR spectra, MIP based on several function monomers (before and after the removal of template) are shown in the following Figures 2, 3, 4, and 5.
By infrared spectroscopy (FTIR), which is used to characterize the structure of the characteristic molecular imprinted polymer content for MET, the beam appears at 1537 cm−1 for ʋC = C stretching and 3330, 3303, 3288 cm−1 for NHNH stretching and 2,947 cm−1. For CH stretching, when compared with FTIR after removal, the disappearance of MET shows an extended band ʋC = C indicating removal of MET and formation of the molecular imprinted polymer.
Infrared spectroscopy (FTIR) is used to diagnose the composition of the molecular imprinted polymer of MET drug, as it is observed by the above diagram showing the beam at 1579 cm-1 for the expansion of C = C, 3415, 3386 cm -1 for the stretching of NH NH, 2827, and 2945 cm−1 for ʋCH stretching which is indicative of drug presence, when compared with the FTIR graph before and after drug removal; MET shows disappearance of ʋC = C band, C = H stretching indicating drug MET removal and molecular fingerprint polymer formation.
Scanning electron microscope (SEM)
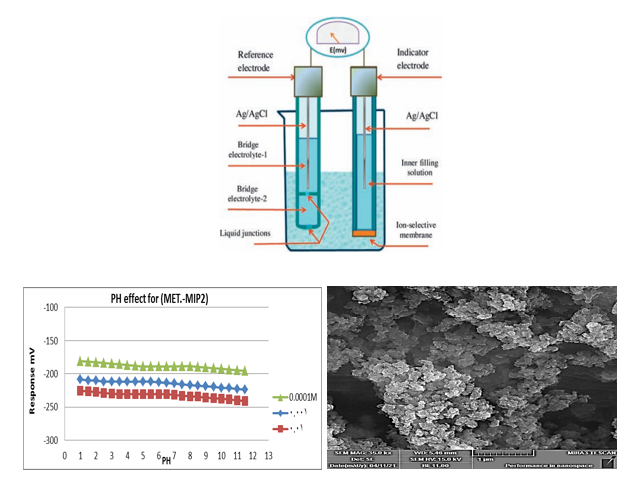
SEM will be utilized to determine the thickness, structure, and surface distribution of the pore membrane. SEM examination revealed that the molecular imprinted polymer has a tightly organized and normal pore structure on the surface and in cross-section, which acts as the interface sites. Several publications have demonstrated that, due to the shape and nature of the porous pores, a molecular impressed membrane of this kind identifies and transports the template molecule efficiently as shown in Figures 6 and 7 the SEM for ( MIP -MET-methyl methacrylate) and (MIP - MET-acryl amide ) after and before washing.
Construction of ion-selective electrodes
By construction of Ion-Selective Electrodes, as shown by Mahajan et al. (12), electrode body building and immobilization were achieved. The solution of metformin (0.1) M was filled as an internal solution in the glass tube. Membrane was preferred tube immersed in a standard solution of (0.1) M naproxen for at least three hours before measurements representing membrane electrode stipulations.
Preparation of pharmaceutical samples
A suitable weight was taken for the production of 100 mL solutions in order to extract the powder of pharmaceutical samples from tablets using a pestle and mortar to grind the tab casting. Appropriate amount of methanol (CH3OH) was utilized for dissolved pharmaceutical samples and completed in a volumetric methanol flask with a magnetic agitator for more than 30 minutes. Instead, the material was screened using 0.07 m cellulose filter paper to prepare and receive levofloxacin concentrations of 1x10–3 M and 1x10–4 M.
Results and discussion
One of the most commonly used voltage sensors is ion selective electrodes (ISE). Such measurements were used in laboratory research, industry, process management, physiological, and environmental monitoring. Membranes that reacted to concentration analysis by generating ions could be controlled by a chemical reaction using an ion selective electrode. The two main types of membrane electrodes were ionic-sensitive selective electrodes and selective molecular electrodes used to evaluate molecular analytes. The fundamental goal of selective ion electrodes is to transmit electrical current from metals to liquids. In metals, electrical current is carried by electrons, whereas in liquids, electrical current is carried by ions. For each electrochemical process, conductivity measurements can be performed in one of these types of galvanic cells, electrolysis, or electrical analysis. This type of cell must be in contact with the solution on both sides of the cell membrane, and some ISE connections are accessible on one side of the membrane. The following is the standard cell composition:
outer ref I test solution I membrane I internal ref.
or
outer ref I test solution I ion selective electrode
The current passing through the electrolytic cell must be zero depending on this condition that the cell is designed in accordance with the basic design rule for electrolytic cells (Figure 8).
Metformin was used as a template, methyl methacrylate was used as a monomer, ethylene glycomethacrylate delayed (EGDMA) was used as a cross-linking agent, and benzoyl peroxide was used as an initiator to make MIP. Plasticizers play a crucial role in the ISE membrane. When a plasticizer is employed as a membrane solvent, compatibility with polymer and other membrane materials offers a homogenous membrane state, and the practical use of an ISE membrane should be banned since the electrode output will be altered over time. A PVC matrix is used to construct two electrodes. Dioctyl phthalate (DOP) as a plasticizer, based on met-MIP (Membranes L1, L2), has been observed. It defines the set of correlation coefficients, linear detection limit range, and age (day). The outcomes are shown in the Table 1.
Effect of pH
The effect of pH on the electrode potentials for (MET) selective membrane electrodes was studied by measuring the e.m.f. of the cell in (MET) solutions at three different concentrations (1×10-4, 1×10-3 and 1×10-2) M in which the pH ranged from 1.0-11.0. The pH adjusted by adding appropriate amounts of hydrochloric acid and/or NaOH solution is shown in Figure 11 and Table 2. At pH values less than 1.5 or in very high acidity, the electrode response has been increased rather irregularly. This may be due to the fact that the electrode response to H+ activities as well as metformin ions and in an alkaline solution (pH greater than 10) has been decreased, which may be attributed to the decrease in the solubility of metformin.
Response Time
Response time is defined as the required time to attain steady potential in the electrode within range of ±1 mV of the final equilibrium value. It has been noticed that the response time value for higher concentrations is less than that of low concentration because the access to the equilibrium state in high concentration is shorter than the low solutions. This proves the response time depending on the concentration of metformin. The average response time (t 95% =) of the metformin membranes is listed in Table (3) below.
Selectivity measurement by separation solution method
Potentiometric selectivity coefficients have been achieved by Separation Solution Method with using metformin concentrations ranging (10-1-5×10-5) M and interfering substances (methyl paraben, propyl paraben, tri sodium citrate); the potentiometric measurement of selectivity coefficients have been done by the equation below:
Log K pot= [(EB − EA)/ (2.303RT/z F)] + (1 –zA/zB) log aA
EA, EB; zA, zB; and aA, represent the potentials, charge numbers, and activities for the primary A and interfering B ions, respectively, at aA = aB.
The results obtained for coefficients of selectivity and interfering ions were listed in Tables 4 and 5, and the selectivity versus the species studied are represented in Figures 12 and 13.
Interference studies
Match potential method (MPM) for selectivity measurement
MPM is used for the selectivity coefficients (Kpot A , B) to determine the electrodes. They are associated with two ions whatever their charge is, since MPM theory is dependent on diffusing electrical layers on both sides (aqueous, interface membranes). So, it does not depend on Nicolsky-Eisenman equation. For the MPM, the selectivity coefficients for equal charge ions (i.e., ZA = ZB) are indicated as the ratio of the standard and interfering ion concentrations in aqueous solutions through the selective surface of the osmotic membrane, neutral ions pass. The selectivity coefficients of different charge ions (i.e. ZA # ZB) not only represented the standard and interfering ions quantities permeating the membrane surface (as a function), but also described the standard ion concentrations within the initial reference solution and the value delta EMF. This method uses the following equation to determine the selectivity coefficient:-
Kpot A,B = a′A -aA/aB
Kpot A, B =the selectivity coefficient, a′A= act as the primary ion A activities, aB=interfering ions. The results are shown in Table 6; the coefficients of selectivity were computed through the interfering ion concentration which gave a potential difference as much as the amount induced due to the increase in the concentration of the primary ion.
We can notice from the data in the Table (6) that there is no overlap between metformin and the interfering ions.
Standard Addition Method (SAM) calculations
During this process, two metformin electrodes at concentrations (10-3 and 10-4) M were applied in this following Equation:
Cu=Cs/10ΔE/s(1+Vu/Vs) - (Vu/Vs)
Where: Cu= the unknown solution concentration; Cs= the standard solution concentration; Vu= the volume of unknown solution; Vs= the volume of standard solution; S= the slope of electrode; E1= electrode potential (mV) in the sample solution; and E2= electrode potential (mV) after the addition of the standard.
They are shown in Tables 7 and 8, and RE% & RSD % for each technique are listed in Tables 9 and 10.
Multiple Standard Addition Method (MSA) Calculations
The measurement in this method were achieved by using two solution of MET applied with concentration (1x10-3and1x10-4) M. The calculations were done by plotting the antilog E/S (Y-axis) against volume of standard naproxen (X-axis). Figures 14, 15, 16, and 17 show that the results of MET concentrations were calculated via the electrodes-based MET-MIP1+DOP, MET-MIP2+DOP, and Table (10) represents the results to these electrodes. The calculation of volume (V) mL and results depended on the equation below:
E=E˚ +S log (C+X Vs/Vu)
Where: S= slope; Vs= the volume of added standard; Vu= the volume of unknown; C= the concentration of an unknown; and X= the concentration of added standard.
Titration Methods (Titrimetry)
These methods include the detection of the endpoint of titrations by depending on the technique of volumetric analysis where the concentrations of the reactants often change gradually; then a large shift takes place in the potential of electrode. The titration curves of metformin at (1×10-3 and 1×10-4) M with a ligand solution [phoshomolybdic acid] (PMA) and results for RSD %, Rec % and Erel % for all electrodes are listed in Table 11.
Applications of pharmaceuticals
Ion selective electrodes were used to assess metformin in pharmaceuticals based on molecularly imprinted polymers. These ISEs were tested using different potentiometric methods. Solutions for metformin at concentrations 1×10-3 and 1×10-4 M were prepared, and Erel%, Rec%, RSD% were determined for metformin in pharmaceutical samples. The obtained results are represented in Tables 12, 13, 14, and 15.
Conclusion
Metformin is used as a template for molecularly imprinted electrode sensors (MIP), and methyl methacrylate (MMA) and acrylamide (AM) are used as monomers in different plasticizers. Using a DOP plasticizer-based electrode, excellent MIP tests with high sensitivity, moderate selectivity, robust static response, long-term stability, and application across a wide pH range were produced. The objective of the building electrodes is to be utilized in the pharmaceutical analysis to determine metformin levels.
Acknowledgements
It’s my pleasure to thank all the staff of Department of Chemistry, College of Science, University of Baghdad, Baghdad, Iraq, for their help.
-----------------------------------------------------------------
How to cite this article: Shams Aws Ismaeel, Yehya Kamal Al-Bayati. Determination of trace metformin in pharmaceutical preparation using molecularly imprinted polymer based pvc-membrane. Eurasian Chemical Communications, 2021, 3(11), 812-830. Link: http://www.echemcom.com/article_138176.html
-----------------------------------------------------------------
Copyright © 2021 by SPC (Sami Publishing Company) + is an open access article distributed under the Creative Commons Attribution License(CC BY) license (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
