Document Type : Original Research Article
Authors
Department of Chemical Engineering, Mahshahr Branch, Islamic Azad University, Mahshahr, Iran
Abstract
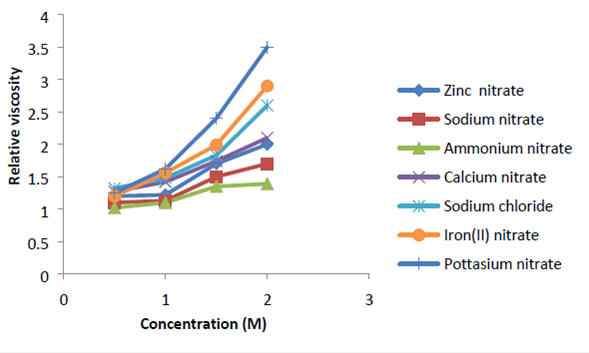
In this study, cleaning of organic-fouled reverse osmosis membranes with direct osmosis high salinity (DO-HS) method by nitrate salts as draw solution was investigated. Synthetic feed water including alginate and natural organic matter were used as model organic foulants. Nitrate salts were selected as draw solution which has not been used for DO-HS cleaning until now. Physicochemical properties of these salts were investigated systematically and related to the DO-HS cleaning performance. The results revealed that the zinc nitrate (1.5 M) draw solution may generate high osmotic pressure more than NaCl. Also, the DO-HS cleaning by zinc nitrate as draw solution was found to be quite effective in cleaning reverse osmosis membranes (cleaning efficiency up to 110%) fouled by gel-forming hydrophilic organic foulants, such as alginate. The effect of physicochemical factors investigated included salt dose, salt type, organic foulants composition and cleaning contact time. It was found that the membrane needed to be cleaned for about 10 min because of the appearance of all foulants on membrane surface. Most importantly, DO-HS cleaning appeared to be an effective cleaning method, and found to be useful for membrane-based advanced wastewater reclamation, where a large fraction of the organic foulants is hydrophilic.
Graphical Abstract
Keywords