Document Type : Original Research Article
Authors
Pharmaceutical Chemistry Department, College of Pharmacy, University of Mosul, Mosul, Iraq
Abstract
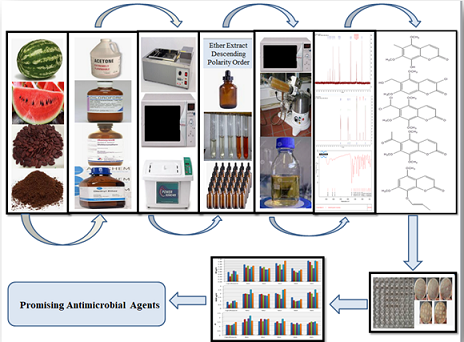
In this study, four different solvents were utilized (acetone, chloroform, dichloromethane, and ether), with three extraction approaches as motion, ultrasonic bath-, and microwave oven-fostered maceration and also, this was performed in three modalities: non-serial, serial ascending-, and descending-design polarity. Among the thirty extracts resulting from the mentioned approaches and modalities, only an ether extract from the descending design polarity was chosen to extract our novel coumarins. This choice is based on the phytochemical examination performed. Extracted coumarins’ structure identification was achieved via gathering and interpretation of the spectral data to those found in the chemical guide. The isolated chemicals’ antimicrobial potential was assessed in vitro via broth-micro dilution technology. The targeted pathogen strains were: six strains of standard pathogenic aerobic gram negative bacteria, four strains of anaerobic bacteria, as well as two strains of fungi. In addition to one strain that is a non-pathogenic intestinal flora. In this assessment, the references used were Ciprofloxacin, Metronidazole, and Nystatin for the aerobic G-, anaerobic, and fungi, respectively. The five novel isolates all indicated antimicrobial potential, but RA4 still exceeded the others. All the tested chemicals exhibited bactericidal and fungicidal potential against all the tested strains of pathogenic bacteria and fungi, based upon PF readings of more than four. On the other hand, RA4 and RA2 were the only two that revealed bacteriostatic effects on the normal flora. Finally, this study concluded the possibility of utilizing these isolated compounds as a source for creating novel antimicrobials.
Graphical Abstract
Keywords
Main Subjects
Introduction
The universal rise in the outbreak and fatality rates related to multidrug-resistant microorganisms is a growing problem worldwide that necessitates the emergence of new therapies [1,2]. This resistance arises when the microbes possess or gain the capacity to bypass the mechanisms which antimicrobials use to combat them. Antimicrobial-resistant infections are more refractory to curing, and they can recur, causing considerable trouble [3,4]. Despite the fact that synthetic antimicrobials have been licensed in many countries, the use of natural chemicals obtained from animal, plant, or microbial sources has piqued the interest of many researchers [5,6]. These chemicals have demonstrated promise in combating antibiotic resistance in microbial infections [7]. Plant-derived chemicals have shown the most promise in fighting microbial infections out of all the available possibilities [5,8,9].
Watermelon fruit is recognized to possess modest calories while being extremely nutrient-dense. Also, it’s rich in dietary fibers, citrulline, and lycopene. It is found to carry various bioactive chemicals, including polyphenols and flavonoids, besides vitamins, notably A and C vitamins, as well as minerals, particularly potassium [10,11]. Watermelon seeds, which are usually discarded, are considered a source of valuable natural bioactive phytochemicals, including alkaloids, cardiac and cyanogenic glycosides, coumarins, flavonoids, oxalate, phytosterols, phenols, saponin, steroids, tannins, and terpenoids [12–14].
Coumarins are naturally occurring chemicals having aromatic properties [15,16]. They were benzopyrones that can be recognized in a variety of therapeutic plants [17–19]. These secondary plant metabolites are present in various parts of plants, including leaves, flowers, stems, roots, and seeds [20,21]. They exhibit a wide range of therapeutic potential, including anti-bacterial [22], anti-viral [23], anti-fungal [24], anti-oxidant [25], anti-cancer [26], anti-hypertensive [27], anti-diabetic [28], anti-inflammatory [29], hepatoprotective [30], anti-coagulant [31], and many other potentials[32,33]. The study of coumarins isolated from watermelon seeds and their potential as antimicrobial agents is a precedent of its kind, as it has not been previously addressed by researchers, despite the evaluation of the antimicrobial activity of extracts from watermelon seeds [34].
The objective of this study is to isolate coumarins from Citrullus lanatus seeds and investigate their in vitro antimicrobial properties, in addition to their safety against normal flora. This goal was accomplished by achieving the following targets: (A) Seeds extraction utilizing four different solvents: acetone, chloroform, dichloromethane, and ether. Extraction was performed using three approaches: motion, microwave oven-, and ultrasonic bath-fostered maceration. Non-serial, serial ascending-and descending-organized polarity were three patterns utilized for each approach. (B) Performing a qualitative phytochemical examination on the extracts. (C) Separating coumarins from the picked sample. (D) Analyzing the chemical backbones of the coumarins which have been isolated, and ultimately, (E) Using a broth dilution manner to evaluate the antimicrobial potential of the isolated coumarin against specified bacterial and fungal strains, in addition to its safety against the non-pathogenic bacteria.
Materials and methods
Sigma-Aldrich and Tokyo Chemical Industry supplied the solvents, chemical substances, as well as microbiological cultures employed in this study. Sisco Research Laboratories Pvt. Ltd. provided the silica gel (mesh size 100-200). Standard fungal, as well as bacterial strains were provided by Microbiologics®. The fruit was bought from a local market in Mosul and then, botanically confirmed by University of Mosul’s College of Agriculture and Forestry specialists. The isolated coumarins’ λ max, as well as their IR spectra was determined utilizing Bruker-Alpha ATR and Varian UV/ Visible. The 1H NMR, as well as 13C NMR spectra of the isolated chemicals was detected on a Bruker Analytische Messtechnik GmbH (300 MHz), employing DMSO-d6 as a solvent.
Plant material proceeding
Every watermelon in the collected batch (164 kilograms) was wholly washed with hydrant water, then by filtered water, and chopped longitudinally into four sections with a sharp knife. The collected seeds were dehydrated in the shadow at room temperature for fourteen days before being grounded with a kitchen grinder and then, sieving mesh was used to produce a fine powder (276 grams). This powder was kept in a tightly closed jar in the fridge for the next stage [35].
The Extraction approach
Four different solvents were utilized to extract the powdery seeds including acetone, chloroform, dichloromethane, and ether. There were three approaches utilized: motion-fostered maceration (MFM), microwave oven-fostered maceration (MOFM), and ultrasonic bath-fostered maceration (UBFM). Extraction was performed in three patterns for every approach: non-serial, serial-ascending, and descending-organized polarity. The powder was extracted in a serial pattern using the initial solvent in the order of polarity. The extracted admixture was filtrated, then the filtrate was subsequently displayed for phytochemical screening; meanwhile, the remnant was extracted via the solvent following the first one in the order of application. In a similar manner, these series were used for the 3rd and 4th solvents [36].
Extraction and isolation [36]
Exactly 200 grams of the grounded seeds were extracted using MPM in a serial-descending pattern. The sequence of solvents was (acetone, chloroform, dichloromethane, and ether). Two litters of the 1st solvent were used, and then the admixture filtrate and the resultant precipitate were allowed to dry before the addition of two litters of the 2nd solvent in the pattern. This sequence continues until reaches the last solvent in the order. Then the extract of the latter solvent was allowed to evaporate by a reduced-pressure rotary evaporator. The crude product mixture (12.86 grams) was agitated in 128.6 milliliters of NaOH (1 M) for 50 minutes at 50°C, and then filtrated. The drop wise addition of hydrochloric acid (1 M) in an ice bath soured the intense-yellow color filtrate; the addition was halted when the color of the solution was faded. The mixture was kept in the fridge for 24 hours to allow the precipitation process to be accomplished, and then the resulting precipitate was filtered and weighted (98.786 milligrams).
In order to find out the number of separated coumarins, a small quantity of the obtained powder in 2 milliliters of ether has been used to produce spots onto the TLC plates. The obtained spots have been mobilized by a mobile phase composed of chloroform: acetone (4:1), then the isolated points were fixed using ultraviolet light (366 nanometers). According to the findings of three trials, the existence of five products was confirmed. The isolation process of these five products was achieved through a gravity chromatography column employing admixtures of ether: ethyl acetate in a tendency ratio beginning at 9:1 and terminating at 1:9, utilized as mobile phases, while the stationary phase was silica gel. Five products, symbolized as RA1-RA5, were recognized, each of which appeared like a single spot on the TLC at various mobile phases.
Spectroscopic data interpretation and physical characteristics of the isolated chemicals RA1-RA5
The data of FTIR, 1H-NMR, 13C-NMR, and physical characteristics regarding the isolated novel coumarins are recorded in Tables 4-7, respectively.
Depiction of chemical backbones for RA1-RA5
By interpreting and comparing the separated coumarins’ spectroscopic data, which were listed above, to those described in the literature, it was revealed that the separated chemicals have a simple coumarin nucleus substituted with two methoxy-groups at positions 5 and 7. Since this backbone is termed limetin [40–43] the separated chemicals RA1-RA5 can be classified as derivatives of this simple coumarin. Based on the already mentioned chemical reference as well as readings of their FTIR, 1H-NMR, and 13C-NMR spectra, the chemical backbones of these five novel-isolated natural coumarins, as displayed in Figure 1, were certified. The IUPAC names of our isolated RAs are: 8-Hydroxy-6-methyllimetin (RA1), (E)-8-(Prop-1'-en-1'-yloxy)limetin (RA2), 3-Chloro-6-(hydroxymethyl)limetin (RA3), 6-(Dichloromethyl)-8-methoxylimetin (RA4), and 6-Acetyl-8-methoxylimetin (RA5).
Microbiological investigation
The ability of the five isolates (RA1, RA2, RA3, RA4, and RA5) to suppress the growth of invasive six aerobic gram-negative and four anaerobic bacterial strains, as well as two types of fungi, was tested. Moreover, the safeness of all isolates against normal gut flora was also examined using E. coli (BAA-1427), as a non-pathogenic bacterial cell.
Evaluation of potential against aerobic gram-negative bacterial strains
This potential was examined using a broth-dilution manner. Mueller-Hinton broth (MHB) was employed here as an environment to promote growth, with Ciprofloxacin (Cip) serving as a control, as well as sulfinylbismethane (DMSO), which is used as a negative qualifier in this famed technique. Two milliliters of the determined isolate with a concentration of 100 milligrams per milliliter were dehydrated first, and then the residue was subsequently measured. In brief, a stock solution was made by mixing 7.5 milligrams of residue with 5 milliliters of DMSO. Then, autoclaved distillate water was employed as a solution-thinner to generate a panel of thirteen two-fold dilutions having marked concentrations of between 1024 and 0.25 micrograms per milliliter. The before-incubation mixture was comprised of the following: The MHB (three milliliters), inoculant calibrated at 0.5 McFarland with (0.2 milliliters) of autoclaved distillate water, as well as a fixed concentration (one milliliter) were transferred into a marked test tube. Following a twenty-four-hour incubation period at 37°C, an ocular examination regarding the growth of bacteria was carried out. The preceding experimental stages were reiterated employing diluted amounts of 0.05, 0.5, 1, or 4, relying upon whatever concentration exhibited insignificant bacterial multiplication. The final stride was to determine the initial micro-biological parameter, which was called the Minimum Inhibitory Concentration (MIC) and was expressed in micrograms per milliliter. The other microbiological parameter, known as the Minimum Bactericidal Concentration (MBC) was examined, as well. The latest was achieved via incubating (three milliliters) of MHB with (0.5 milliliters) of the second row’s diluted proportions. Ultimately, via dividing the readings of both MBC as well as MIC over one another, the third parameter, recognized as Potency Factor (PF), was determined for every isolate against each utilized bacterium. To enhance the accuracy, the approach was used three times.
Evaluation of potential against anaerobic bacterial strains
The approach utilized to determine the potential of the detected isolates towards anaerobic bacterial pathogens was similar to that utilized to determine the potential against aerobic pathogenic bacteria, while there were several obvious distinctions. The parameters that have been altered involved employing DMSO as a qualifier, Brucella-agar enriched with the blood of sheep (five percent) as a growth-enhancing medium, and Metronidazole (Metro) as a control. Furthermore, the incubation was achieved over two days at 37 °C in a reservoir containing an anaerobic midst (N2 80 percent, CO2, and H2 10 percent each), an indicator of anaerobic type, and the catalyst used was the metal palladium.
Evaluation of potential against infectious fungal strains
The approach utilized to estimate the isolates’ antifungal potential differed slightly from that utilized to investigate their potential against aerobic bacterial strains. The amended parameters involved the employment of Nystatin (Nyst) as a control, Sabouraud-dextrose broth as a growth-enhancing medium, and incubation at 30 °C for a two-day period of time.
Results and discussion
Relative safety and accessibility to a wide variety of natural products support their extensive use among a diversity of communities to profit from their therapeutic capabilities. The separation and structural interpretation of phytochemical constituents are two of the major hitches that researchers encountered in phytochemistry. These hitches are growing more sophisticated as trials are made to correlate the existence of specific moieties in the chemical backbones of separated compounds with different biological properties [44,45].
Map of the natural products
The grounded seeds of Citrullus lanatus were extracted using four different solvents: acetone, chloroform, dichloromethane, and ether. In this extraction, three approaches were utilized such as MPM, UPM, and DPM. Each approach was tested in three different patterns: non-serial, serial ascending-, and descending-organized polarity. This procedure resulted in thirty extracts being examined for the presence of the specified 1ry and 2ry phytochemical constituents. The findings reported in Tables 1-3 demonstrated that simple coumarins were found in all extracts as a consequence of the extraction approaches and the employed patterns. Coumarins are often extracted using a variety of solvents; this is primarily determined by the nature of the functional units linked to the coumarin nucleus and the amount of the extracting solvent employed [46]. Coumarins are distinguished structurally by the availability of lactone moiety, which can easily be hydrolyzed into derivatives of cis-cinnamic acid that are water-soluble when attacked by a potent nucleophile such as sodium hydroxide. When these derivative products are reacted with potent acid, the original coumarins are regenerated [47]. In the existing research, the previous chemical pattern was utilized as a strategy for natural coumarins’ isolation. The ether extract from serial MPM of descending pattern was picked to separate its coumarin constituents. This choice was attributed to the absence of specific phytochemical constituents in this isolate, such as alkaloids, fixed oils, flavonoids, and tannins which could hinder the separating technique [48].
Inspection of antimicrobial potential
Across civilized history, there have been continuing struggles between infectious creatures and human beings, which have on occasion manifested in troubles due to the inability of the present remedies to overcome resistant contagion-caused germs. Because antimicrobials will still be utilized as a tool to enhance this fight, the quest for efficient medicines necessitates intensive research to detect novel antimicrobial products, particularly those derived from natural sources [18,49,50]. In this aspect, several research studies on the antimicrobial properties of many simple coumarins have been published [51,52]. However even so, the association between their structural properties and antimicrobial potential has received scant attention in the literature. Depending on the current findings, simple coumarins with increased lipophilicity may have greater antimicrobial efficacy than those with lower ones [53]. This is consistent with the findings seen in the latter Tables.
Anti-Aerobic Gram-Negative Bacterial (anti-AG-ve Bac) Potential
The separated products' anti-AG-ve Bac potential was evaluated against a variety of typical harmful bacteria, comprising Escherichia coli (Esch. coli, ATCC 25922), Salmonella typhi (Sa. typhi, ATCC 6539), Shigella dysenteriae (Sh. Dysenteriae, ATCC 13313), Haemophilus influenzae (Haemo. Influenza, ATCC 49247), Klebsiella pneumonia (Kl. Pneumonia, ATCC 700603), and Pseudomonas aeruginosa (Pseudo. Aeruginosa, ATCC 27853) in addition to Escherichia coli (Esch. coli, BAA-1427) as the non-pathogenic one.
Based on the findings in Table 8, which were depicted graphically in Figure 2, the researchers concluded that the separated coumarin-based scaffolds have the following order of potential: RA4, RA2, RA1, RA3, and RA5. According to this order, it was established that the anti-AG-ve Bac potential grew as the lipophilicity of the group occupied position six rose [54,55]. Among those five isolated coumarin-based scaffolds, RA4 indicates a more comparable anti-AG-ve Bac potential to Ciprofloxacin than the others. The authors postulate that the existence of the two chloride atoms in the substituted group (CHCl2) at position number six can function as an acceptor in H-bonding, aside from its medium-sized atomic radius [56]. The previous two characteristics permit the benzopyrone structure to be introduced and adhere perfectly via the pockets of the target that possess lipophilic or lipophobic features [57]. As for the RA5, the presence of the (COCH3) group at position 6 allows for the formation of more H-bondings, increasing its hydrophilicity and decreasing its anti-AG-ve Bac potential [54]. Finally, what was concluded from the evaluation of these isolated chemicals is that they all possess bactericidal potential versus the tested bacteria. This finding originated from relying on PF values which were lower than four [58].
On the other hand, regarding the safety of the isolated products towards normal gut flora, all revealed much higher safety than Ciprofloxacin against normal flora. The previous fact was deduced from the values of MIC and MABC shown in Table 8 and graphically represented in Figure 3. The safest one is RA4, followed by RA2, RA5, RA3, and RA1. Finally, it is noteworthy to mention that, despite their potential as bactericidal agents against the pathogenic bacteria investigated, RA4 and RA2 displayed a bacteriostatic impact against the assessed normal flora based on the PF values which were higher than 4 [58,59].
Anti-Anaerobic Bacterial (anti-AnABac) Potential
The potential of the five isolated simple coumarins was evaluated as anti-AnABac agents versus four bacterial strains: Clostridium perfringens (Cl. Perfringens, ATCC 13124), Bacteroides fragilis (Ba. Fragilis, ATCC 25285), Prevotella melaninogenica (Pr. Melaninogenica, ATCC 25845), and Fusobacterium necrophorum (Fu. Necrophorum, ATCC 25286). This is done in an anaerobic condition using a Brucella-agar microdilution procedure. According to the findings mentioned in Table 9, which were analyzed diagrammatically in Figure 4, all the isolated products could function as anti-AnABac but still exhibited a lower potential than the control drug. In addition, all the tested products possessed bactericidal potential [55].
Anti-Infectious fungal potential
This potential was examined against two fungal strains, comprising Candida albicans (Cand. Albicans, ATCC 10231) and Aspergillus niger (Asperg. niger ATCC 16888) using a Sabouraud-dextrose broth dilution approach, as mentioned above. As evidenced by the findings in Table 10, which were graphically displayed in Figure 5, RA4 possesses the same anti-fungal potential as Nystatin versus Cand. albicans and a little less towards Asperg. niger. The other four isolated products all revealed lower potential than the control drug to act as antifungals toward the assessed pathogens. The researchers attributed RA4's remarkable potential to the availability of two chloride atoms in the substituted group at position six (CHCl2). Because it is coupled to a highly-conjugated order, this substitution can boost the molecule’s cellular uptake as well as its affinity for the target [55,60]. In addition, according to PF values, all the isolated products revealed fungicidal potential against the tested fungi [61].
Conclusion
The current study proved successful findings in the separation and structural description of newfound five simple coumarin-based scaffolds (limetin-derivatives) obtained from Citrullus lanatus seeds. Furthermore, the antimicrobial study of these isolated simple coumarins revealed their ability to serve as an encouraging backbone for the creation of new antimicrobial medicines thanks to the following findings: their remarkable antibacterial and antifungal potential against the tested bacteria and fungi, their possession of bactericidal and fungicidal potential against the tested pathogens, as well as their relatively high safety towards normal intestinal flora. Finally, despite the fact that all of the tested compounds exhibit antimicrobial potential, RA4 is still superior to others in terms of effectiveness and safety against non-pathogenic bacteria.
Acknowledgements
The authors are thankful for the support and for the facilities offered by the University of Mosul/College of Pharmacy, which contributed to enhancing the quality of this study. The authors would still like to thank Drs. Reem Nadher Ismael, Sara Firas Jasim, and Sarah Ahmed Waheed for their contributions to the quality of this study.
Ethical issues
The scientific committee of the Pharmaceutical Chemistry Department was approved this work.
Competing interests
We have no conflicts of interest to disclose.
Authors’ contributions:
All authors contributed toward data analysis, drafting and revising the paper and agreed to responsible for all the aspects of this work.
Orcid:
Rahma Mowaffaq Jebir: https://www.orcid.org/0000-0002-8712-4713
Yasser Fakri Mustafa: https://www.orcid.org/0000-0002-0926-7428
-------------------------------------------------------------------------------
How to cite this article: Rahma Mowaffaq Jebir*, Yasser Fakri Mustafa. Novel coumarins isolated from the seeds of Citrullus lanatus as potential antimicrobial agents. Eurasian Chemical Communications, 2022, 4(8), 692-708. Link: http://www.echemcom.com/article_147423.html
-------------------------------------------------------------------------------
Copyright © 2022 by SPC (Sami Publishing Company) + is an open access article distributed under the Creative Commons Attribution License(CC BY) license (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
