Document Type : Original Research Article
Authors
Department of Chemistry, College of Education for Pure Science, University of Mosul, Mosul, Iraq
Abstract
The study included partially purified of polyamine oxidase (PAO) from sheep's brain tissue by dialysis and ion exchange chromatography using DEAE-cellulose techniques. Isoenzymes (I and II) were obtained with specific activities 3.876 and 2.856 units/ mg of protein and purification folds 11 and 8 times respectively compared with crude enzyme.
The specific activity of PAO I was better than PAO II, thus we considered it for following studying. The optimal conditions were 40ͦ C and pH=9. Some of properties of partially purified PAO I enzyme were studied and the specificity was found to be superior to the substrate, where it was given the highest activity when using spermidine. The results showed the presence of cupper ion at a concentration of 0.1437 mg / mL in PAO I using an atomic absorption. The presence of the coenzyme conjugate flavin adenine dinucleotide (FAD) was also detected by using UV-Vis spectrum from (325-430) nm. The inhibitory effect of carbamazepine on the activity of homozygous PAO I was studied, and it was found that there was a decrease in the activity of the enzyme at different inhibitor concentrations. The type of inhibition of the drug was also studied, and it was found non-competitive, and that the value of the inhibition constant Ki was 1.8 mM. The polyamines in the crude extract of Sheep's brain were diagnosed with HPLC technique, and the results of the analysis indicated the presence of spermine, spermidine, cadavarin, diamino propane and hexylamine as well as the presence of other polymines.
Graphical Abstract
Keywords
Main Subjects
Introduction
Polymines are bio-organic compounds of low molecular weight consisting of an aliphatic carbon chain with several amino groups, include Spemine, Spermidine, and Putrescine. They are found in all types of cells of mammals [30]. PAs are natural components of all living prokaryotic and eukaryotic cells [33], where they play a wide range of physiological roles. PA is present as small organic cations inside cells. They are associated with organic anions, nucleic acids, proteins and phospholipids through electrostatic bonds that can cause stability, destabilization or in combination with phenolic compounds [19,34]. Several studies have indicated that cancerous tissues in the breast [23], the kidney, prostate, and bladder [15], pancreas [25] and colon [27] contain high concentrations of polyamine compounds, and its derivatives compared to natural fabric. Another study, describing the distribution of polyamine compounds in 10 regions of the brains of people for different age groups, indicated that PAs are distributed in normal adult brain tissue [30]. A recent study indicated that high levels of polyamine compounds were found in the central nervous system in animals and humans [38].
Polyamine oxidases (PAOs) are single-chain peptide enzymes (monomers) that are important in the biological processes of generating naturally occurring amines [14], as they contribute to the homeostasis of polyamine compounds [8]. These enzymes are found in most tissues of vertebrates and invertebrates and are located mainly in the mitochondria, and play an important role in the vertebrate brain [36]. It is also found in mammals, plants, bacteria and fungi [18]. These enzymes have been studied in different tissues and organs, including the liver and brain of rats, human and rabbit blood serum [28], and blood and aortic plasma in sheep [17], where these enzymes stimulate oxidation of a terminal amine group, free or substituted and the products of the reaction are hydrogen peroxide H2O2 and ammonium ion NH4 + [37].
Materials and methods
Sheep's brain collection
Quarter kilogram of Sheep's brain was obtained from slaughter house in November 2017 in Mosul, Iraq. The brain was free from diseases and stored at – 18 °C until next steps.
Preparation of crude extraction
One hundred gram of sheep brain tissue was taken, crushed using a blender crushing machine for 10 minutes and mixed with sodium phosphate buffer, and the extract was frozen for 24 hours, then left to thaw at room temperature. The process was repeated three times. After that, the mixture was stirred for two hours by an electric motor, subject to cooling in an ice bath, and the mixture was filtered by several layers of gauze, the extract was separated with a chilled centrifuge to dispose insoluble material for 10 minutes at 3000 xg and to obtain a clear filtrate, and volume of the filtrate which represent volume of crude extract was measured.
Determination of protein
Modified Lowry method was used to estimate protein concentration at different stages of enzyme purification. The standard curve was done by using bovine serum albumin [35].
Assay of polyamine oxidase
The activity of PAO was measured using modified Flayeh method [20]. The enzyme activity was estimated by following the decrease in absorbance resulting from reduction of potassium ferrocyanide at the wavelength of 410 nm by using spermine as substrate.
Purification of PAO from the crude sheep's brain
Dialysis
It was the first step of purification using cellophane against sodium-potassium phosphate buffer for 12 hr. at 4 °C and the resulting sample was used for the next step of purification process.
Ion exchange chromatography
The solution enzyme which obtained from previous step was loaded on DEAE-Cellulose resin for purification. In test tubes, 5 mL was collected in each one. The flow rate of elution was 1 mL/min. The filtered protein fractions were followed by measuring the absorbance at a wavelength of 280 nm and the activity of the PAO. The collected enzyme fractions were lyophilized for using in the next steps.
Study of optimum conditions of purified PAO
The effect of temperature on activity of purified enzyme was studied by incubating the enzyme for 10 minutes at different temperatures ranging from (0-70 °C). The effect of the sodium-potassium phosphate buffers at a concentration (20 mM) with pH range (4-10) were investigated.
Specificity of purified PAO I towards different substrates
The specificity of PAO was studied towards the different base materials, which include spermine, spermidine, cadavarin, diamine propane, benzyl amine, hexylamine and butylamine at a concentration of 100 mM substrate.
Determination of copper ion by atomic absorption and FAD by UV-Vis Spectrum
Copper ion associated with purified enzyme was detected using an atomic absorption spectrometer at wavelength 324.8 nm, and UV-Vis at (325-430) spectrum was used for detection of conjugate coenzyme FAD [26].
Inhibition of Purified PAO
Inhibitory effect of Carbamazepine drug on PAO I activity
The effect of carbamazepine on PAO I activity was studied using different concentrations of the drug, ranging between (0.2- 2 mM). This done by incubating 0.2 mL of the enzyme with 0.1 mL of carbamazepine solution at a temperature of 37 °C for a period of 30 minutes, then an enzyme activity was measured [9].
Study of inhibition type
For investigating the type of inhibition, 0.2 mL of purified PAO was incubated with 0.1 mL of carbamazepine (0.8 mM) at a temperature of 37 °C for a period of 30 minutes. The enzyme activity was estimated using different concentrations of spermine ranging between (25-200 mM).
Diagnosis of polyamine compounds in sheep's brain tissue using HPLC technique
Standard samples were prepared for analysis by HPLC apparatus using a diameter separating column 4.6 mm high, 25 cm high, and filled with CH3Si (CH3) 17 silica, which is known as a separation column (C18) [24] as follows:
Preparation of compounds for analyzation
One mL of obtained crude extract was added to 10 mL of 0.2N perchloric acid. Then the sample was incubated in an ice bath for 40 minutes. The centrifugation process was carried out at 4 °C for a period of 20 minutes, after which 0.5 mL of the filtrate was taken, and 1 mL of 2N NaOH solution was added to it with 10 μl of benzoyl chloride, and the mixture was left at the laboratory temperature for 20 minutes. The reaction was stopped by adding 2 mL of saturated solution NaCl and the benzoylamide derivative was extracted by adding 2 mL of diethyl ether solution, then the organic layer was taken and dried by passing nitrogen gas, and the dried product was dissolved with 120 μl of methanol. Standard polyamines (Spm, Spd, DAP, Cadavarin and Hexylamine) were treated in the same way above with a concentration of 1 micromole [11].
Results and discussion
Purification of PAO
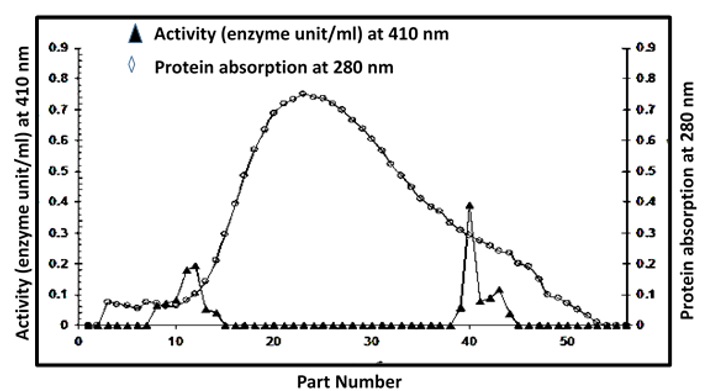
The results shown in Table 1 indicates that the specific activity of PAO enzyme after dialysis step became 1.977 enzyme units/mg protein. That is, the membrane screening process increased the purity of the enzyme by 5.68 times compared to the crude extract. This result is consistent with [5]. The reason for this increase in the specific activity of PAO can be attributed to the elimination of small molecular weight compounds such as some amino acids, peptides and ions that can surround the active site thus reduce the enzyme activity. After applying the sample in the separator column containing the anion exchanger DEAE-Cellulose, the elution indicated the presence of two distinct peaks possessing PAO activity (Figure 1).
Table 1 shows that the first peak (PAO I) appeared at an elution volume (40-75) mL with a specific activity of 3.876 enzyme U/mg protein and 31.89% recovery activity. The second peak (PAO II) appeared at an elution volume (195-225) mL with a specific activity of 2.856 enzyme U/mg protein and 24.30% recovery activity. These results were identical to what was found for the purified PAO from breast milk, where two distinct peaks each possessed activity of PAO, and the specific efficacy of these peaks reached 17.36 and 13.02 enzyme U/mg protein, respectively [4]. The purified cerebrospinal fluid of children revealed the appearance of two distinct peaks, each possessing the activity of the PAO enzyme, and the specific activity of these peaks was 1201.92 and 1157.22 enzyme units/mg protein, respectively [41].
Polyamine oxidases were partially purified from red blood cells hemolysate from atherosclerotic patients as part of the study. Two polyamine oxidase isoenzymes with specific activities of (0.148 and 0.163 x10-3) U/mg protein were discovered [6]. On other hand, one peak of PAO was partially purified from cow’s brain, using DEAE- cellulose with specific activity 6.34 U/mg protein [2].
Study of the optimum conditions of purified PAO
The results in Figure 2 indicate a gradual increase in the enzymatic activity values with increasing temperature, reaching a maximum at 40 °C. The rise in temperature increases the kinetic energy of the enzyme, which increases the enzymatic affinity with the substrate and causes an increase in the speed of the reaction, but at high temperatures, this leads to the enzyme deformation. These results are close to the optimum temperature of the PAO in citrus, which was 36 °C using spermidine, while it gave a lower optimum value of 30 °C using Spm as substrate [44]. The optimum temperature of purified PAO erythrocytes in normal and diabetic women was found to be 37 °C [5]. However, PAO optimal temperature in pregnant mothers' serum was 55ºC [21].
The results in Figure 3 show the highest activity of enzyme at optimum pH 9 using the buffer solution KH2PO4-Na2HPO4. It is known that the energy of each enzyme depends on the nature of the molecular ionization of its component protein, thus it can be said that the vitality of the enzyme depends on the degree of hydrogen ion concentration in the middle of the reaction. The acidic function of the enzyme can change the secondary and tertiary structure of the enzyme molecule or the active site which affects the rate of the enzymatic reaction. Optimum pH of PAO for cancerous colon cells was found to be 9 [21]. But optimal pH of breast milk purified PAO was found 8.6 [4], while in citrus the highest activity was found at pH 8 and 7 using Spm and Spd as substrate respectively [44].
Effect of different substrates on PAO I activity
Table 2 indicates the effect of different substrates (spermine, spermidine, cadavarin, diamine propane, butylamine, hexylamine and benzylamine). PAO I showed highest specificity toward spermidine followed by spermine and then cadavarin. These results are similar to [4], while the maximum specificity was found toward spermine and spermidine respectively of PAO from blood cells of healthy women [5].
Determination of cupper ion and FAD
By using atomic absorption Spectrometer, PAO I showed the presence of a cupper ion at a concentration of 0.1437 mg/mL , which indicates that Cu+ 2 plays a role in enzyme activity. Al-Rubaiie illustrateed the presence of cupper ions with concentrations of 0.1915 and 0.2947 mg/mL in the analogues of PAO in green pepper, while noted the absence of the ion in an enzyme of red blood cells for women with type 1 diabetes [5]. Copper ion is a coenzyme of MAO as well as PAO [10,40].
The obtained results using the UV-Vis spectrum of the partially purified PAO I revealed the presence of coenzyme FAD at wavelengths (325-430 nm) (Figure 4). Some enzymes require the presence of non-protein molecules called conjugate factors [12]. This is in agreement with Vujcic [43] who indicated that PAO contains FAD in all vertebrate tissues, since these enzymatic companions contribute to redox reactions, and they are small organic molecules that are strongly or weakly bound to the enzyme. Al-Lehebe also found that purified PAO from red blood cells are effective for healthy women and diabetics contained FAD [5].
Inhibitory effect of carbamazepine drug on PAO I activity
Carbamazepine is a pharmaceutical drug that contains an amine group. The approved medical uses for this drug have been identified by FDA in the United States and have been epilepsy including partial seizures, generalized tonic-clonic seizures, mixed seizures, trigeminal neuralgia, manic episodes, and mixed seizures of type 1 bipolar disorder [42]. In view of the role played by amine oxidase enzymes in neurological diseases, many studies confirm the high level of these enzymes in such pathological conditions as bacterial meningitis [1], viral meningitis [22], depression and schizophrenia [15]. Therefore, we decided to study the inhibitory effect of this drug on the activity of PAO.
The results indicate that carbamazepine had an inhibitory effect on this enzyme and that the value of IC50 was 0.8 mM, as shown in the Table 3. Therefore, we can say that the inhibitory effect of this drug towards PAO may be related to its synthetic formula containing an amine group that is likely to work in binding to the active site or part of it, or perhaps this amine group that enzyme works on and is present in the composition of substrate.
The results indicated that the inhibition was of the non-competitive type, Km and Km´ were 43.4 mM, while Vmax decreased from 0.145 to 0.120 units/mL, as shown in Figure 5, and inhibition constant Ki was 1.8 mM. Using the drug, amikazine, the inhibition was non-competitive for the purified PAO from green pepper, with a value of Km and Ki were 58.8 and 2 mM respectively [7]. While Al-Abbasi found a competitive inhibition of PAO of people with type 2 diabetes by using metformin with vitamin E. The Km and Ki values were 1.33 and 51 mM respectively [3].
Identification of polyamine compounds present in Sheep's brain extract using HPLC chromatography
Amino acid nutrition is critical for maintaining health and productivity in sheep and goats. Specifically, the amino acid profiles of ruminal microbial protein and animal production protein must be balanced [13].
The results show in Figures 6-10 that obtained by HPLC, refer to standard polyamine chromatogrms. The appearance of peaks at retention times specific to each compound are shown in Table 4.
Figure 11 shows several peaks related to polyamine compounds (spermine, spermidine, cadavarin, dipropane and hexylamine) with concentrations and retention times as indicated in Table 4, while the other peaks may be attributed to the presence of other monoamines or other polymines compounds.
Previous research indicated the presence of Spm, Spd and Put in mammals [32]. Rat liver tissue is very rich in PA compounds, as the levels of spermine, spermidine and putrescine were estimated [11]. Polyamine compounds were diagnosed by using HPLC which indicated the higher levels in RBC than plasma, and spermine level was higher than spermidine and diamino propane levels [5].
There were differences in polyamines concentrations in foods, meat and fish which contain high concentrations of putrescine and spermine and low concentrations of spermidine [22]. It was also found that mushrooms contain higher concentrations of spermidine compared to spermine [30].
Conclusion
In this study, Polyamine oxidase was purified partially from sheep's brain and then characterized. The inhibition of purified PAO I activity by carbamazepine was improved non-competitively. Cupper ion and FAD are existing with PAO peak as a cofactor and coenzyme. The polyamines in the crude extract of sheep brain were diagnosed with HPLC technique.
Acknowledgments
We would like to appreciate and thank Mosul University, College of Education for Pure Sciences, Chemistry Department, for helping and provided that the requirements for conducting this work.
Orcid:
Sarah Abdulelah Younis:
https://orcid.org/0000-0002-0372-0885
Wathba Idrees Ali: https://orcid.org/0000-0003-2066-4122
-------------------------------------------------------------------------------------
How to cite this article: Sarah Abdulelah Younis*, Wathba Idrees Ali. Identification of polyamine compounds and characterization of polyamine oxidase from sheep's brain tissue. Eurasian Chemical Communications, 2021, 3(9), 644-655. Link: http://www.echemcom.com/article_134802.html
-------------------------------------------------------------------------------------
Copyright © 2021 by SPC (Sami Publishing Company) + is an open access article distributed under the Creative Commons Attribution License(CC BY) license (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
